

Technology > Microarrays
At the heart of Akonni’s TruDiagnosis® solution is the TruArray® technology, which utilizes three-dimensional (3D) gel drop microarrays to rapidly screen a sample for hundreds of disease markers at one time, all within a microarray grid the size of your fingernail. Akonni has an exclusive license from Argonne National Laboratory to a robust patent portfolio surrounding the 3D gel drop technology, which is supported by >60 peer-reviewed publications [1]. In addition, Akonni has created its own patent portfolio, which includes improvements to the microarray manufacturing, a novel microfluidic control device, and the hardware/software platform for running and analyzing the microarray tests.
3D gel drop arrays have a sterically-favorable spacing of immobilized molecules throughout the volume of the gel drop, and a 99% aqueous environment surrounding the covalently attached probes. The net result of these attributes is an increase in hybridization efficiency [2], and predictable aqueous-based reaction kinetics versus difficult to implement surface-based reactions. 3D gel drops also have a higher probe immobilization capacity, which increases detection sensitivity up to 100-fold compared with 2D planar arrays [3]. This improved detection sensitivity translates to low-cost instrumentation.
Gel drop arrays also have a corollary manufacturing advantage over some other microarray formats (beads, in situ synthesis, or planar surfaces), in that one-and-the-same manufacturing infrastructure and methodology can be used to produce protein and nucleic acid diagnostic tests. Each gel drop can be considered a ‘nano-test-tube‘, where each individual drop may be loaded with varying concentrations of the capture molecule or with other reaction ingredients to facilitate the biochemical assay. In addition, the co-polymerization methodology can be implemented on unmodified plastic substrates, which eliminates the need for high-priced glass substrates and surface treatments, allowing for significantly lower costs of goods.
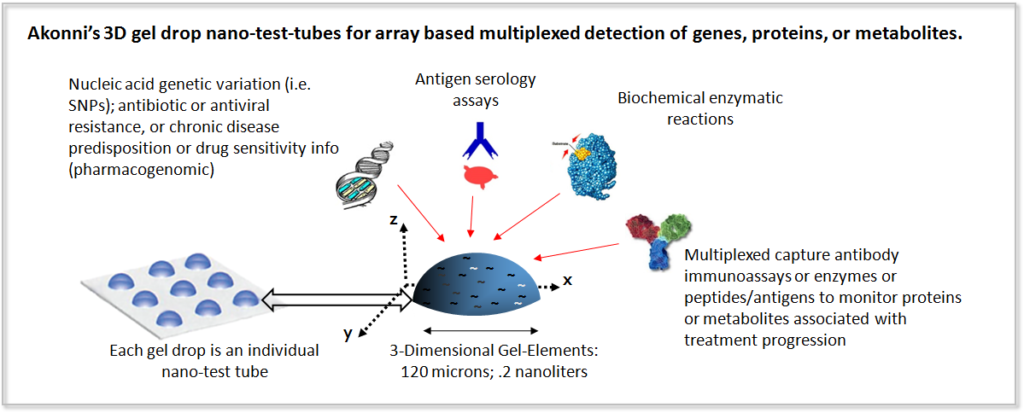
Akonni’s proprietary gel drop microarrays have been shown to be amenable to a plastic substrate, instead of a traditional glass slide. The unique co-polymerization methodology can be implemented on plastic films, which eliminates the need for high-priced glass slides and surface treatments.
Our team has invented a microfluidic test device with an all plastic design, and a plastic film-based substrate, which significantly reduces manufacturing costs versus glass, silicon wafers, or microelectronic chips utilized by competitors. We have patented the manufacturing process for reel-to-reel manufacturing of arrays on film, which will completely change the industry’s cost structure for a multi-panel test, similar to the breakthrough enabled by lateral flow test production methods (i.e. pregnancy, HIV, or drugs of abuse test strips sold in pharmacies today).

The ability to print on unmodified plastic, combined with the valve-less microfluidic approach, enables us to manufacture the Akonni TruArrays in a reel-to-reel format. Reel-to-reel manufacturing could conceivably reduce the cost by one to two orders of magnitude as compared with traditional microfluidic cartridges.
Reel-to-reel assembly is a method of high volume production used predominantly for the assembly of lateral flow strips and flexible film electronics. The virtue of this manufacturing method is that high volume production can be very cost effective. Using standard production equipment, assembly of the devices can be completely automated at very high speeds. In fact, lateral flow strips, manufactured in this reel-to-reel format, often cost less than a dollar.
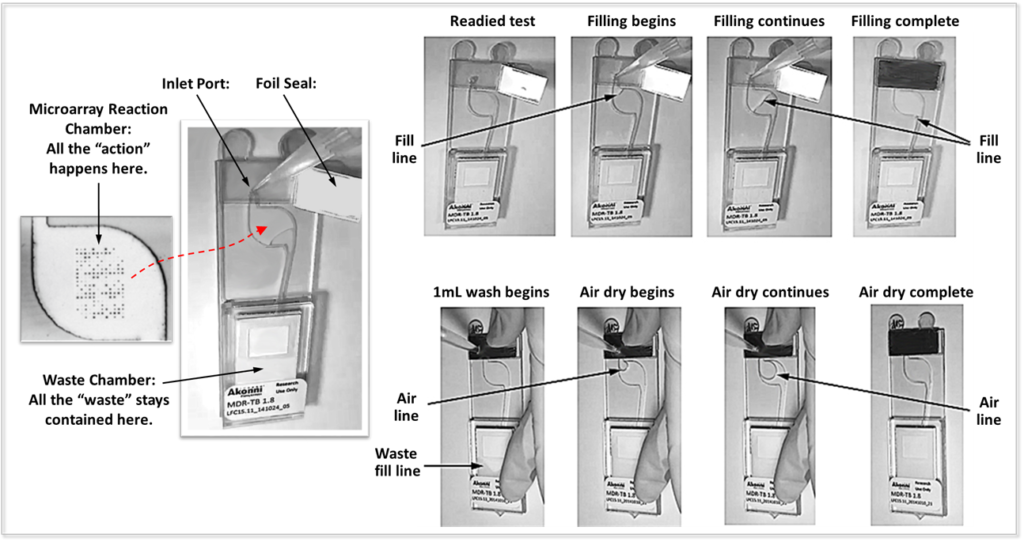
To address the need for detecting multiple targets in a self-contained, low-cost disposable device, Akonni has developed a proprietary, innovative technology that distinguishes Akonni’s TruArrays from other microarray technologies – the integrated microfluidic microarray consumable (IMMC).
The microfluidic innovation of this design is that these closed-amplicon microarray devices do not require valves, which are frequently required and utilized in competing PCR-based microfluidic devices. Furthermore, unlike existing technologies, Akonni’s microfluidic device does not require special hydrophobic coatings or treatments. Instead, we have demonstrated that a proprietary design based on geometry and specialty materials confines the liquid reagents in the microarray chamber until an additional reagent, such as a wash solution, is added.
The unique, simple, yet elegant design of Akonni’s IMMCs allow PCR amplification to occur in the 3D gel drop array chamber of the IMMC, eliminating the need for multiple fluidic transfer steps, and includes an integrated, fully-contained waste chamber, making it a closed system. The waste chamber and the microarray chamber are separated by a microfluidic stop, which confines the reaction mix to the microarray chamber during thermocycling. The design enables the microarray surface to be washed after amplification and hybridization, while maintaining a closed-amplicon system. After the process is complete, the amplified product and reagents flow into the integrated waste chamber, virtually eliminating the risk of PCR contamination.
(1) Source: Pubmed
(2) References
Khrapko, Y. Lysov, A. Khorlin, I. Ivanov, GM, A method for DNA sequencing by hybridization with oligonucleotide matrix, Mitochondrial DNA. (1991).
Stomakhin, V. Vasiliskov, E. Timofeev, D. Schulga,… oligonucleotide microchips: MALDI mass spectrometry identification of 5mers contiguously stacked to…, Nucleic Acids Research. (2000).
(3) References
Guschin, G. Yershov, A. Zaslavsky, A. Gemmell, V, Manual manufacturing of oligonucleotide, DNA, and protein microchips, Analytical Biochemistry. (1997).
Gavin, A. Kukhtin, D. Glesne, D. Schabacker, DP, Analysis of protein interaction and function with a 3-dimensional MALDI-MS protein array, BioTechniques. (2005).
© 2003-2024 Akonni Biosystems, Inc. All rights reserved. Terms and Conditions • Privacy Policy
TruDiagnosis Systems, TruDx Readers, TruArray Tests, TruTip Kits, Primers, Lysis and Binding Buffers, Wash Buffers and Elution Buffers: For Research Use Only. Not for use in diagnostic procedures. No claim or representation is intended to provide information for the diagnosis, prevention or treatment of a disease.

Based on its recent analysis of the microarrays in molecular diagnostics (MDx) market, Frost & Sullivan recognizes Akonni Biosystems with the 2017 North American New Product Innovation Award.
Akonni’s integrated MDx system, TruDiagnosis®, is revolutionizing the point-of-care molecular testing market by enabling target detection from a variety of sample types. TruDiagnosis® is powered by TruArray®, a patented 3D gel-drop microarray technology for sample screening that instantly indicates the presence of disease markers in real time.
Download: